If Z is a compressibility factor, van der Waals equation at low
$ 19.99 · 4.5 (684) · In stock

Solution For If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 1: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 2: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 3: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 4: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

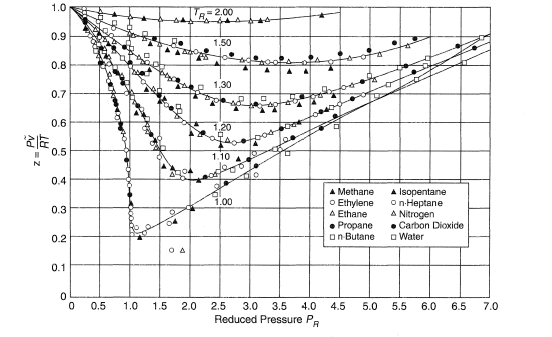
Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt
GAS LAW

66. If z is the compressibility factor, van der Waals equation low

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange
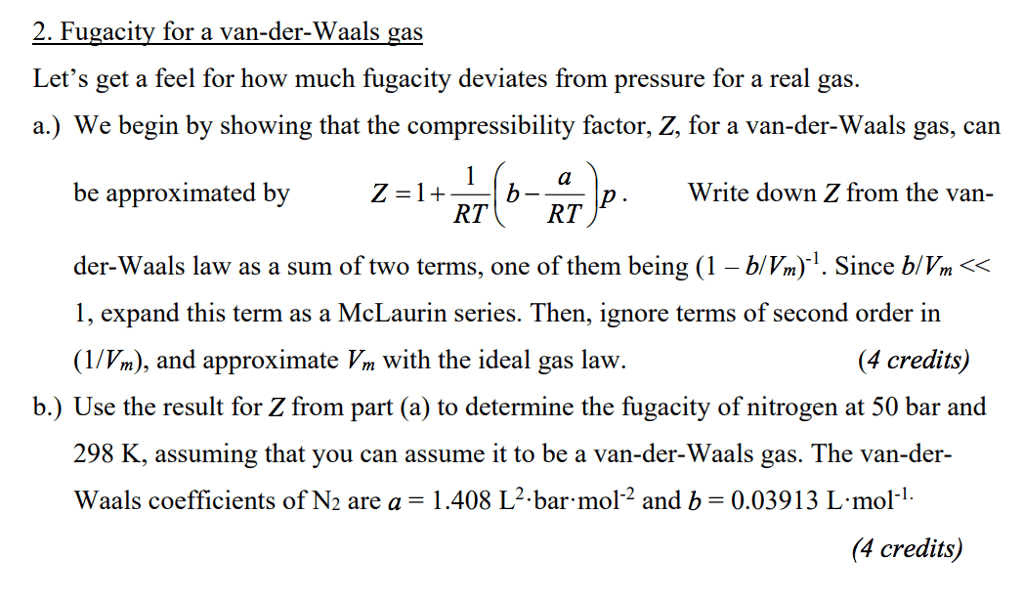
OneClass: At low pressures the compressibility factor for a Van der Waal's gas is given by Z-1+[b- (a

If Z is a compressibility factor, van der Waals' equation at low
Thermo] Derivation of compressibility factor vs reduced pressure